
Iodine production methods
Iodide is found in small amounts in seawater, algae, shellfish, and fish liver. It is extracted either from natural ores, such as sources in Chile, or from underground brines. In the air blowing method, iodide is converted into iodine and separated by an air stream. In the ion exchange method, iodine is absorbed by resin and then separated and precipitated using chemicals. In the adsorption method on activated carbon, iodine is adsorbed by charcoal and recovered with an alkaline solution. The pH of the solution and the concentration of alkali have a significant impact on this process.
Iodine is present in seawater in small amounts in the form of iodide ions, and its concentration reaches about 50 mg per ton of seawater. It is also formed in seaweed (with a concentration of 900 ppm), shellfish, and fish liver.
Iodine extraction is carried out by two main methods:
Extraction from natural ores: In this method, iodine is obtained from a type of ore containing iodine. In Chile, as the world’s largest producer, iodine is extracted from a specific type of ore that has a concentration of about 400 mg per liter.
Recovery from underground brines: In this method, iodine-containing brines are extracted by drilling wells deep into the ground and then iodine is separated from it. Brine from deep wells or brine extracted from oil wells contain a maximum of 150 mg per liter of iodine.
Methods of Iodine Production from Underground Brine
Blowing-out Method
This method takes advantage of the high volatility of iodine and is suitable for treating brine at high temperatures. First, sand and impurities in the brine are removed by sedimentation, and an acid and an oxidant are added to convert iodide ions into iodine molecules. Then, this brine is sprayed from the top of a stripping tower so that the iodine contained in it is separated from the brine by the air flow and evaporated. In the next step, the iodine-containing air is directed to the absorption tower, where an absorbent material absorbs and concentrates the iodine.
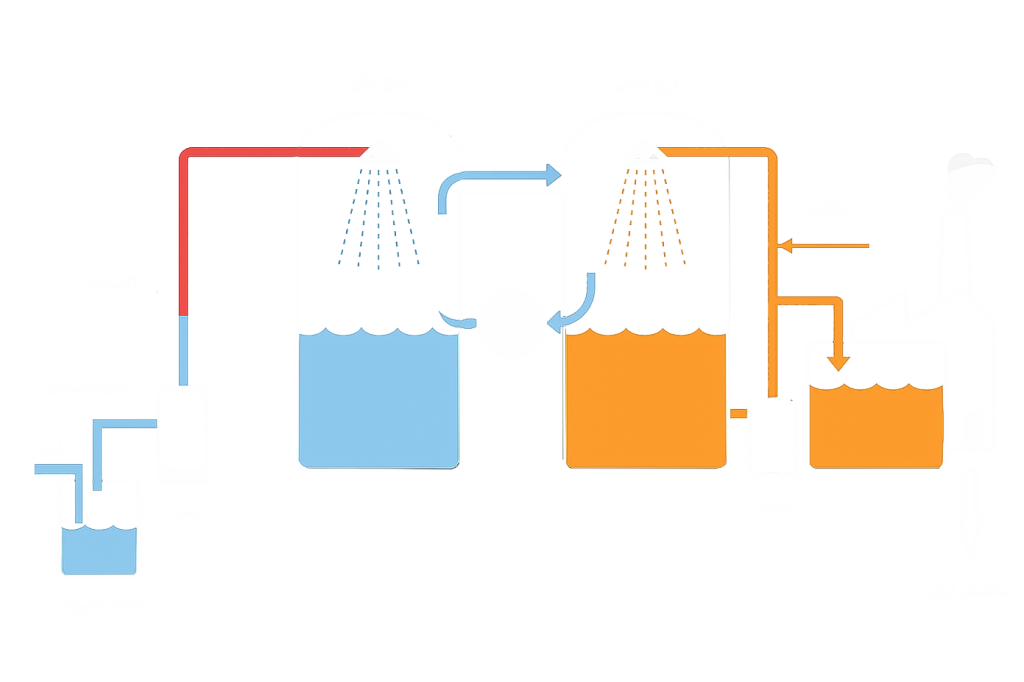
And then the solution containing the absorbed iodine is sent to the production building to be converted into solid iodine in the production process.
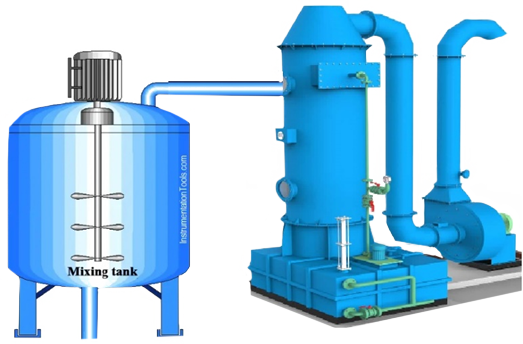
Ion Exchange Method
In this method, sand and impurities are removed from the brine by sedimentation and filtration, and an oxidant such as chlorine gas or sodium hypochlorite is added to convert the iodide ions into iodine molecules. The brine is then passed through an adsorption column filled with ion exchange resin to absorb the iodine. The resin, which has completely absorbed the iodine, is placed in a desorption column to remove the iodine with a sulfuric acid (H2SO3) solution. Chlorine gas is then added to the desorbed solution to precipitate iodine crystals.
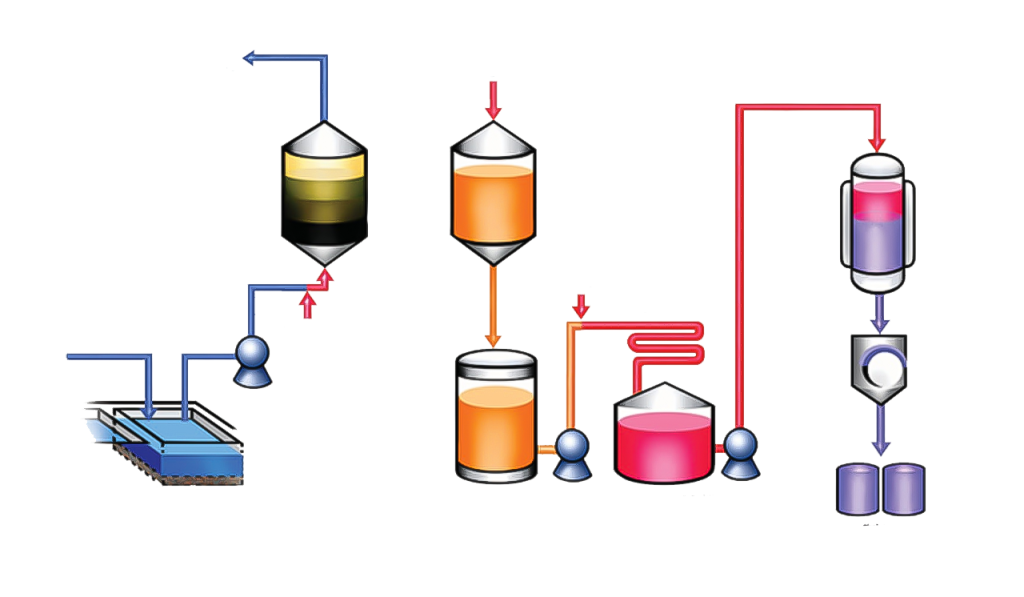
Activated Carbon Adsorption Method
The activated carbon adsorption method is used to extract iodine from dilute aqueous solutions, such as natural brines and bitter waters. This method was one of the first methods used to extract iodine. In this method, iodine is adsorbed by granular activated carbon, and after saturation of the carbon, iodine is removed from the activated carbon using a sodium hydroxide solution. In this method, the brine is first acidified and then oxidized with an oxidant. This process releases molecular iodine into the solution. Passing this solution over the activated carbon causes iodine to be adsorbed onto the charcoal. To recover the adsorbed iodine, a sodium hydroxide or sodium carbonate solution is passed through the charcoal, and finally the washed solution is oxidized again with acidic sulfuric acid and with sodium nitrite, which leads to iodine precipitation.
The adsorption of iodine from dilute solutions onto activated carbon can be well correlated with the Freundlich equation, and the adsorption kinetics almost follows a first-order irreversible reaction. The results have shown that the pH of the solution in the adsorption stage and the concentration of sodium hydroxide solution in the desorption stage have the greatest effect on the adsorption and desorption rates of iodine, respectively. In addition, although some of the activated carbon is lost in the adsorption and desorption processes, its adsorption and desorption properties are still similar to the original activated carbon after several regenerations.