Physical Properties of Iodine
Iodine is a non-metallic element belonging to the halogen group (Group 17) in the periodic table. This element is distinguished among the halogens due to its unique physical properties. Iodine appears as a solid with a dark gray to blackish-purple color and has a metallic luster. When heated, iodine sublimates easily into a purple-colored gas.


Iodine is never found in nature in its elemental form and does not concentrate enough to form independent mineral deposits. The first ionization potential of iodine atoms is relatively low, so iodine readily combines directly with many elements to form iodides.
Some technical and chemical properties of iodine are listed in the table below:
| Property | Value |
|---|---|
| Atomic Number | 53 |
| Element Symbol | I |
| Discoverer | Bernard Courtois (1811) |
| Atomic Weight | 126.9 |
| Density | 4.93 g/cm³ (at 20℃) |
| Melting Point | 113.6℃ |
| Boiling Point | 184.4℃ |
| Electron Configuration | [Kr] 4d 10 5s 2 5p 5 |
| Phase at Standard Conditions (STP) | Solid |
Applications of Iodine
Generally, iodine is used in the food industry (as a salt additive, food stabilizer, and animal feed), pharmaceutical industry (disinfectants, X-ray contrast agents, and iodine tablets), chemical industry (as a catalyst in acetic acid and polymer production, in the production of high-purity metals, chemical fertilizers, and other iodine compounds), electronics industry (in manufacturing LEDs and LCDs), and more.
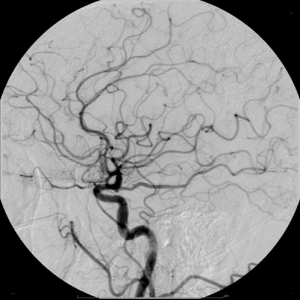
X-ray Contrast Agent
Iodine has a high ability to absorb X-rays. Utilizing this property, iodine-based contrast agents are administered intravenously to improve and enhance the clarity of X-ray and CT images (such as lung CT scans).

Disinfectants and Antimicrobial Agents
Due to its inherent antimicrobial properties, iodine is used in disinfectants and antimicrobial agents. Povidone-iodine (Betadine) is a strong disinfectant from the iodophor group and is widely used in first aid. Because iodine is absorbed slowly through soft tissue, it provides longer-lasting disinfectant effects and is suitable for longer surgeries.
Iodine tincture is another disinfectant commonly used in hospitals and medical applications.

Food Additive and Salt Supplement
Iodine is a key component of thyroid hormones, which regulate and boost the body’s metabolism. This element is essential for the growth of living organisms. Iodine increases the metabolism of fats, sugars, and proteins and helps burn body fat, thereby contributing to obesity prevention. Insufficient iodine intake leads to iodine deficiency disorders, resulting in conditions such as impaired growth. To prevent such diseases, iodine is added to animal feed and salt.
