Introduction: The Formation of Liquid Droplets in the Atmosphere
Most people are usually taught a simplified version of how clouds form. For example, it is often said that clouds form when water vapor condenses into liquid water droplets. However, the actual process is somewhat more complex. To form water droplets in the atmosphere, water vapor must condense onto a series of tiny particles called aerosols. In other words, water vapor needs particles of an appropriate size to transition into liquid form. These tiny particles are known as cloud condensation nuclei (CCN).
In clean air, where no CCN are present, water vapor can become supercooled and spontaneously form droplets after about 5 to 6 hours at around -13°C [1,2]. Aerosol particles must either be water-soluble (such as various salt particles) or have a sufficiently large radius (greater than 0.1 micrometers) and a hydrophilic surface to allow condensation [3].
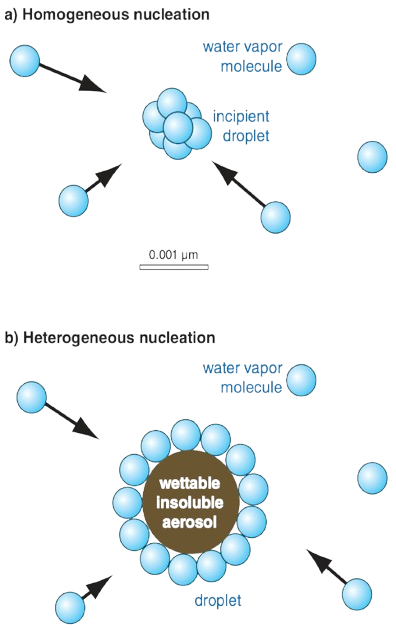
To form a raindrop, solid aerosols serve as condensation sites for water vapor. In cloud seeding, small particles of silver iodide are introduced into the atmosphere to induce cloud formation and precipitation. Aerosols naturally originate from dust, soot, salt crystals, or molecules released by plants [4]. Additionally, sulfuric acid and ammonia molecules can aggregate in the atmosphere to form new aerosol particles. Modeling studies indicate that more than half of cloud droplets form on aerosols generated within the atmosphere. Importantly, the composition of aerosol particles is less critical than their size; ultimately, aerosols grow to about 100 nanometers or larger to become effective CCN [5,6].
Naturally, CCN can form from the aggregation or oxidation of pollutant gases in the presence of sunlight. Pollutant gas condensation and coagulation cause aerosols to grow rapidly to sizes between 0.01 and 1 micrometer, suitable for water vapor condensation. Over oceans, CCN may consist of sulfur-containing compounds like sulfate and sulfuric acid, which are produced from gases such as dimethyl sulfide and methanesulfonic acid emitted by phytoplankton.
Besides sulfur, researchers have recently identified another element associated with marine algae that influences atmospheric reactions over the ocean. Iodine has been introduced as a substance highly prone to aerosol particle formation. These particles can grow over time to become cloud condensation nuclei, positively affecting cloud formation and precipitation. It is noteworthy that all living organisms contain some amount of iodine. For example, in humans, iodine supports the production of hormones that regulate metabolism and aid brain and bone development during pregnancy.
Following the Chernobyl accident in 1986, potassium iodide tablets were distributed in an effort to mitigate the harmful effects of radiation on health. Although iodine has been studied in aqueous solutions for centuries, scientists have only taken significant steps in the past two decades to understand its gaseous phase impact on Earth’s atmosphere. Recent studies show gas-phase chemical reactions where iodine converts to iodic acid (HIO3), which acts catalytically in atmospheric particle formation [7].
Currently, it is established that iodine is the only element that, after release from the Earth’s surface, does not leave the atmosphere but can cycle back to the gas phase through redox reactions while still in particulate form. This means iodine can act as an important catalyst in cloud formation [8].
While sulfuric acid, methanesulfonic acid, and nitric acid require ammonia or dimethylamine for aerosol formation, iodine can form aerosols by oxidizing abundant organic molecules present in the atmosphere. The nucleation rate of iodic acid (HIO3) exceeds that of sulfuric acid. Atmospheric sulfuric acid levels have decreased due to pollution controls, whereas iodine emissions have tripled since the 1950s and continue to rise due to factors such as increasing ozone concentration and thinning Arctic ice [10,11]. Recently, it has become a hot topic that air pollution control in cities causes warming by reducing sulfuric acid concentrations [9].
New Findings on the Positive Impact of Iodine on Cloud Condensation and Precipitation
In recent years, the “Cosmics Leaving Outdoor Droplets” (CLOUD) project, a collaboration among 21 institutions at the CERN particle accelerator in Geneva (see figure below), has studied how iodine-containing vapors form aerosol particles and how these grow into cloud droplets [6,9,12]. CLOUD experiments, conducted in one of the world’s cleanest laboratories, employ various instruments to analyze the physical and chemical properties of atmospheric gases and particles [5]. The results have been corroborated by field measurements at the Maido Observatory on Reunion Island, located east of Madagascar. The team spent ten weeks using scientific instruments to characterize molecules on the island and found real-world data consistent with laboratory findings [7].

A 2022 CLOUD study [13] revealed two chemical mechanisms by which iodine influences cloud formation. In one, iodine forms aerosols without sulfuric acid. Phytoplankton convert iodate in ocean water to iodide, likely using it as a simple mineral antioxidant to protect their cell walls. Surface iodide reacts with atmospheric ozone, producing molecular iodine and volatile iodine-containing organic compounds (I2, HOI, VOI) that release into the atmosphere, photolyze, and oxidize to form iodine oxides (IxOy) or iodic acids (HIO3, HIO2) [11]. Molecular iodine converts readily to iodic acids without ultraviolet radiation or daylight, allowing rapid aerosol formation [5]. These iodine oxide and iodic acid clusters provide sites for rapid accumulation of non-volatile organic compounds. Volatile organic compounds (C1-C5) from phytoplankton oxidize to form polyhydric alcohols (tetrols), multifunctional carbonyls (glyoxal), low-volatility organic acids, and highly hygroscopic alkylaminium salts. This aggregation causes initial iodine oxide clusters to grow to about 1 micrometer (Aitken particles), becoming CCN essential for vapor condensation and precipitation. A significant portion of iodine associated with these growing particles recycles to the gas phase, acting as a “catalyst” for nucleation and aerosol production in marine atmospheres [11].
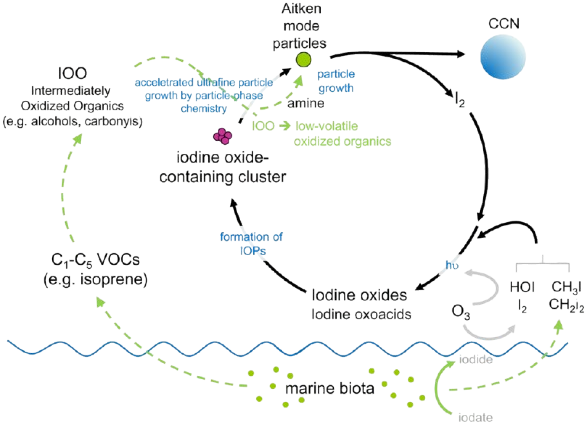
Further field studies confirmed the dominant role of HIO3 in nucleation and CCN formation [10]. HIO3 has been detected in marine coastal air, polar boundary layers, various continental sites, and the lower troposphere. Several precursors to HIO3 have been proposed, including hydrated iodine atoms, hydrated IO radicals, OIO radicals, and larger iodine oxides (I2O3, I2O4, I2O5). The 2023 discovery of the HIO3 formation mechanism, published in Nature Chemistry [10], revealed iodine’s catalytic role in aerosol formation. The reaction pathway involves:
(R1) IOIO + O3 → IOIO4
(R2) IOIO4 + H2O → HIO3 + HOI + (1)O2
Another mechanism involves interaction between iodine oxoacids and sulfuric acid to form CCN and water droplets [9]. Sulfuric acid requires a stabilizer like ammonia in urban areas, but ammonia is scarce in pristine environments such as oceans. CLOUD researchers found that iodic acid can enhance sulfuric acid ion nucleation. Remarkably, iodous acid can replace ammonia by acting as a base and accepting a proton from sulfuric acid to form a neutral dimer, which is even more effective. Thus, iodine can increase particle production efficiency from sulfuric acid by 10 to 10,000 times compared to ammonia [6,9,14], playing a critical role in CCN production.
Summary
Clouds are not gas collections but assemblages of tiny water droplets and/or ice crystals, mostly in the troposphere. Rain clouds usually contain larger water droplets, appearing darker and more likely to precipitate. These droplets and ice crystals form by condensation or deposition of water vapor onto condensation nuclei (CCN). In other words, water vapor must be saturated and condense onto particles. Rising moist air cools, saturates, and condenses water vapor onto CCN, leading to cloud growth and precipitation [15].
Recent research on molecular iodine (I2) shows that iodine molecules contribute positively to precipitation by forming cloud condensation nuclei, facilitating cloud seeding and rainfall. Thus, gaseous iodine not only has no negative effect on precipitation but also plays a positive role. Iodine aids in forming primary aerosol particles, growing them into CCN, and thereby promoting condensation and rainfall, much like silver iodide used in cloud seeding. Unlike sulfuric acid, iodine does not require ammonia for aerosol formation, significantly accelerating nucleation and CCN growth.
For further studies and up-to-date research on iodine’s positive impact on cloud condensation and precipitation, see the recommended videos and articles published since 2020.
Video:
https://videos.cern.ch/record/2751109
Research Papers about the effect of iodine on the formation of cloud condensation nuclei
- Huang, Ru-Jin, et al. “Heterogeneous iodine-organic chemistry fast-tracks marine new particle formation.” Proceedings of the National Academy of Sciences32 (2022): e2201729119.
- Rörup, Birte, et al. “Temperature, humidity, and ionisation effect of iodine oxoacid nucleation.” Environmental science: atmospheres5 (2024): 531-546.
- He, Xu-Cheng, et al. “Role of iodine oxoacids in atmospheric aerosol nucleation.” Science6529 (2021): 589-595.
- Xavier, Carlton, et al. “Role of Iodine-Assisted Aerosol Particle Formation in Antarctica.” Environmental Science & Technology17 (2024): 7314-7324.
- Baccarini, Andrea, et al. “Frequent new particle formation over the high Arctic pack ice by enhanced iodine emissions.” Nature communications1 (2020): 4924.
- de Jonge, Robin Wollesen, et al. “Natural marine precursors boost continental new particle formation and production of cloud condensation nuclei.” Environmental Science & Technology25 (2024): 10956-10968.
- He, Xu-Cheng, et al. “Iodine oxoacids enhance nucleation of sulfuric acid particles in the atmosphere.” Science6676 (2023): 1308-1314.
- Finkenzeller, Henning, et al. “The gas-phase formation mechanism of iodic acid as an atmospheric aerosol source.” Nature Chemistry1 (2023): 129-135.
- Zhang, Rongjie, et al. “Critical role of iodous acid in neutral iodine oxoacid nucleation.” Environmental Science & Technology19 (2022): 14166-14177.
- Wan, Yibei, et al. “Probing key organic substances driving new particle growth initiated by iodine nucleation in coastal atmosphere.” Atmospheric Chemistry and Physics Discussions2020 (2020): 1-29.
- Tham, Yee Jun, et al. “Direct field evidence of autocatalytic iodine release from atmospheric aerosol.” Proceedings of the National Academy of Sciences4 (2021): e2009951118.
- Kirkby, Jasper, et al. “Atmospheric new particle formation from the CERN CLOUD experiment.” Nature Geoscience11 (2023): 948-957.
- Shen, Jiali. “Oxidation of Dimethyl Sulfide and Iodine in the Marine Atmosphere, and their Contribution to new Particle Formation.” (2023).
- Shen, Jiali. “Oxidation of Dimethyl Sulfide and Iodine in the Marine Atmosphere, and their Contribution to new Particle Formation.” (2023).
- Ning, An, et al. “Overlooked significance of iodic acid in new particle formation in the continental atmosphere.” Proceedings of the National Academy of Sciences31 (2024): e2404595121.
- Zhang, Ying, et al. “Iodine oxoacids and their roles in sub-3 nm particle growth in polluted urban environments.” Atmospheric Chemistry and Physics3 (2024): 1873-1893.
- Engsvang, Morten, Haide Wu, and Jonas Elm. “Iodine clusters in the atmosphere I: Computational benchmark and dimer formation of oxyacids and oxides.” ACS omega29 (2024): 31521-31532.
- Zhang, Ying, et al. “Iodine oxoacids and their roles in sub-3 nanometer particle growth in polluted urban environments.” EGUsphere2023 (2023): 1-39.
- Baccarini, Andrea, et al. “Iodine Drives New Particle Formation in the Central Arctic Ocean.” AGU Fall Meeting Abstracts. Vol. 2020. 2020.
- Wan, Yibei, et al. “Chemical characterization of organic compounds involved in iodine-initiated new particle formation from coastal macroalgal emission.” Atmospheric Chemistry and Physics23 (2022): 15413-15423.
Report references
[1] Climate Intervention, National Academies Press, Washington, D.C., 2015. https://doi.org/10.17226/18988.
[2] J.G. Hudson, Cloud condensation nuclei, J. Appl. Meteorol. (1993). https://doi.org/10.1175/1520-0450(1993)032<0596:CCN>2.0.CO;2.
[3] R. Stull, 7.2: Nucleation of Liquid Droplets – Geosciences LibreTexts, Libr. Geosci. (2022). https://geo.libretexts.org/Bookshelves/Meteorology_and_Climate_Science/Practical_Meteorology_(Stull)/07%3A_Precipitation_Processes/7.02%3A_Nucleation_of_Liquid_Droplets (accessed May 2, 2025).
[4] K.R. Barry, T.C.J. Hill, E.J.T. Levin, C.H. Twohy, K.A. Moore, Z.D. Weller, D.W. Toohey, M. Reeves, T. Campos, R. Geiss, G.P. Schill, E. V. Fischer, S.M. Kreidenweis, P.J. DeMott, Observations of Ice Nucleating Particles in the Free Troposphere From Western US Wildfires, J. Geophys. Res. Atmos. (2021). https://doi.org/10.1029/2020JD033752.
[5] Goethe University — Climate research: rapid formation of iodic particles over the Arctic – more clouds could cause ice to melt faster, (n.d.). https://www.goethe-university-frankfurt.de/97794506/Climate_research__rapid_formation_of_iodic_particles_over_the_Arctic___more_clouds_could_cause_ice_to_melt_faster (accessed May 2, 2025).
[6] X.C. He, Y.J. Tham, L. Dada, M. Wang, H. Finkenzeller, D. Stolzenburg, S. Iyer, M. Simon, A. Kürten, J. Shen, B. Rörup, M. Rissanen, S. Schobesberger, R. Baalbaki, D.S. Wang, T.K. Koenig, T. Jokinen, N. Sarnela, L.J. Beck, J. Almeida, S. Amanatidis, A. Amorim, F. Ataei, A. Baccarini, B. Bertozzi, F. Bianchi, S. Brilke, L. Caudillo, D. Chen, R. Chiu, B. Chu, A. Dias, A. Ding, J. Dommen, J. Duplissy, I. El Haddad, L.G. Carracedo, M. Granzin, A. Hansel, M. Heinritzi, V. Hofbauer, H. Junninen, J. Kangasluoma, D. Kemppainen, C. Kim, W. Kong, J.E. Krechmer, A. Kvashin, T. Laitinen, H. Lamkaddam, C.P. Lee, K. Lehtipalo, M. Leiminger, Z. Li, V. Makhmutov, H.E. Manninen, G. Marie, R. Marten, S. Mathot, R.L. Mauldin, B. Mentler, O. Möhler, T. Müller, W. Nie, A. Onnela, T. Petäjä, J. Pfeifer, M. Philippov, A. Ranjithkumar, A. Saiz-Lopez, I. Salma, W. Scholz, S. Schuchmann, B. Schulze, G. Steiner, Y. Stozhkov, C. Tauber, A. Tomé, R.C. Thakur, O. Väisänen, M. Vazquez-Pufleau, A.C. Wagner, Y. Wang, S.K. Weber, P.M. Winkler, Y. Wu, M. Xiao, C. Yan, Q. Ye, A. Ylisirniö, M. Zauner-Wieczorek, Q. Zha, P. Zhou, R.C. Flagan, J. Curtius, U. Baltensperger, M. Kulmala, V.M. Kerminen, T. Kurtén, N.M. Donahue, R. Volkamer, J. Kirkby, D.R. Worsnop, M. Sipilä, Role of iodine oxoacids in atmospheric aerosol nucleation, Science (80-. ). 371 (2021) 589–595. https://doi.org/10.1126/science.abe0298.
[7] Atmospheric iodine could contribute to more cloudy skies | Popular Science, (n.d.). https://www.popsci.com/environment/iodine-cloud-formation-ozone/ (accessed May 3, 2025).
[8] Iodine accelerates formation of cloud condensation nuclei in the atmosphere | Press and Public Relations, (n.d.). https://press.uni-mainz.de/iodine-accelerates-formation-of-cloud-condensation-nuclei-in-the-atmosphere/ (accessed May 2, 2025).
[9] X.C. He, M. Simon, S. Iyer, H. Bin Xie, B. Rörup, J. Shen, H. Finkenzeller, D. Stolzenburg, R. Zhang, A. Baccarini, Y.J. Tham, M. Wang, S. Amanatidis, A.A. Piedehierro, A. Amorim, R. Baalbaki, Z. Brasseur, L. Caudillo, B. Chu, L. Dada, J. Duplissy, I. El Haddad, R.C. Flagan, M. Granzin, A. Hansel, M. Heinritzi, V. Hofbauer, T. Jokinen, D. Kemppainen, W. Kong, J. Krechmer, A. Kürten, H. Lamkaddam, B. Lopez, F. Ma, N.G.A. Mahfouz, V. Makhmutov, H.E. Manninen, G. Marie, R. Marten, D. Massabò, R.L. Mauldin, B. Mentler, A. Onnela, T. Petäjä, J. Pfeifer, M. Philippov, A. Ranjithkumar, M.P. Rissanen, S. Schobesberger, W. Scholz, B. Schulze, M. Surdu, R.C. Thakur, A. Tomé, A.C. Wagner, D. Wang, Y. Wang, S.K. Weber, A. Welti, P.M. Winkler, M. Zauner-Wieczorek, U. Baltensperger, J. Curtius, T. Kurtén, D.R. Worsnop, R. Volkamer, K. Lehtipalo, J. Kirkby, N.M. Donahue, M. Sipilä, M. Kulmala, Iodine oxoacids enhance nucleation of sulfuric acid particles in the atmosphere, Science (80-. ). (2023). https://doi.org/10.1126/science.adh2526.
[10] H. Finkenzeller, S. Iyer, X.C. He, M. Simon, T.K. Koenig, C.F. Lee, R. Valiev, V. Hofbauer, A. Amorim, R. Baalbaki, A. Baccarini, L. Beck, D.M. Bell, L. Caudillo, D. Chen, R. Chiu, B. Chu, L. Dada, J. Duplissy, M. Heinritzi, D. Kemppainen, C. Kim, J. Krechmer, A. Kürten, A. Kvashnin, H. Lamkaddam, C.P. Lee, K. Lehtipalo, Z. Li, V. Makhmutov, H.E. Manninen, G. Marie, R. Marten, R.L. Mauldin, B. Mentler, T. Müller, T. Petäjä, M. Philippov, A. Ranjithkumar, B. Rörup, J. Shen, D. Stolzenburg, C. Tauber, Y.J. Tham, A. Tomé, M. Vazquez-Pufleau, A.C. Wagner, D.S. Wang, M. Wang, Y. Wang, S.K. Weber, W. Nie, Y. Wu, M. Xiao, Q. Ye, M. Zauner-Wieczorek, A. Hansel, U. Baltensperger, J. Brioude, J. Curtius, N.M. Donahue, I. El Haddad, R.C. Flagan, M. Kulmala, J. Kirkby, M. Sipilä, D.R. Worsnop, T. Kurten, M. Rissanen, R. Volkamer, The gas-phase formation mechanism of iodic acid as an atmospheric aerosol source, Nat. Chem. (2023). https://doi.org/10.1038/s41557-022-01067-z.
[11] J.C. Gómez Martín, T.R. Lewis, M.A. Blitz, J.M.C. Plane, M. Kumar, J.S. Francisco, A. Saiz-Lopez, A gas-to-particle conversion mechanism helps to explain atmospheric particle formation through clustering of iodine oxides, Nat. Commun. (2020). https://doi.org/10.1038/s41467-020-18252-8.
[12] How iodine-containing molecules contribute to the formation of atmospheric aerosols, affect climate | ScienceDaily, (n.d.). https://www.sciencedaily.com/releases/2021/02/210208085521.htm (accessed May 3, 2025).
[13] R.J. Huang, T. Hoffmann, J. Ovadnevaite, A. Laaksonen, H. Kokkola, W. Xu, W. Xu, D. Ceburnis, R. Zhang, J.H. Seinfeld, C. O’Dowd, Heterogeneous iodine-organic chemistry fast-tracks marine new particle formation, Proc. Natl. Acad. Sci. U. S. A. 119 (2022). https://doi.org/10.1073/pnas.2201729119.
[14] H. Zu, S. Zhang, L. Liu, X. Zhang, The vital role of sulfuric acid in iodine oxoacids nucleation: impacts of urban pollutants on marine atmosphere, Environ. Res. Lett. 19 (2024) 014076. https://doi.org/10.1088/1748-9326/ad193f.
[15] S.T. Siems, Cloud seeding, in: Encycl. Earth Sci. Ser., 2013: p. 92. https://doi.org/10.1007/978-1-4020-4399-4_63.
[1] An aerosol is any tiny solid or liquid particle suspended in the air.
[2] Cloud Condensation Nuclei
[3] Aitken nucleus refers to aerosol particles that grow with density to a size larger than 1 micrometer.
